
Thus, the molar mass of bilirubin can be expressed as 584.73 g/mol, which is read as “five hundred eighty four point seventy three grams per mole. The division sign (/) implies “per,” and “1” is implied in the denominator. The mass in grams of 1 mole of substance is its molar mass. Thus, the molar mass of bilirubin can be expressed as 584. Element name 80 Symbol Boron Electronegativity. Do not round them further when performing calculations. All average masses are to quantities, and subject to significant figure rules. The relative atomic mass is the same as the molar mass (except molar mass is in g/mol). The Periodic Table of the Elements 1 18 Hydrogen 1 H 1.01 2.1 be treated as measured 2 Average relative masses are 2001 values, rounded to two decimal places. Do this by looking up sodium (Na) on the periodic table. For example, find the mass of one mole of sodium. The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g. Example 1: Find the Molar Mass of an Element. For example, the molar mass of Ba(OH) 2 requires the sum of 1 mass of Ba, 2 masses of O, and 2 masses of H: The molar mass of Ba(OH)2 requires the sum of 1 mass of Ba, 2 masses of O, and 2 masses of H: 1 Ba molar mass:īecause molar mass is defined as the mass for 1 mol of a substance, we can refer to molar mass as grams per mole (g/mol). One mole of Al atoms has a mass in grams that is numerically equivalent to the atomic mass of aluminum. In formulas with polyatomic ions in parentheses, the subscript outside the parentheses is applied to every atom inside the parentheses. For elements with no stable isotopes, the mass number of the isotope with the longest half-life is in parentheses. adenosine (C 10H 13N 5O 4), a component of cell nuclei crucial for cell divisionīe careful when counting atoms.barium sulfate (BaSO 4), used to take X rays of the gastrointestional tract The first step for calculating molar mass is to identify all the elements in a given molecule and write their atomic masses using the periodic table.Practice problems assess your knowledge of this chemistry process. What is the mass of 1 mol of each substance? Test your ability to find the molar mass of compounds and convert moles to grams in this multiple choice quiz and worksheet combo.
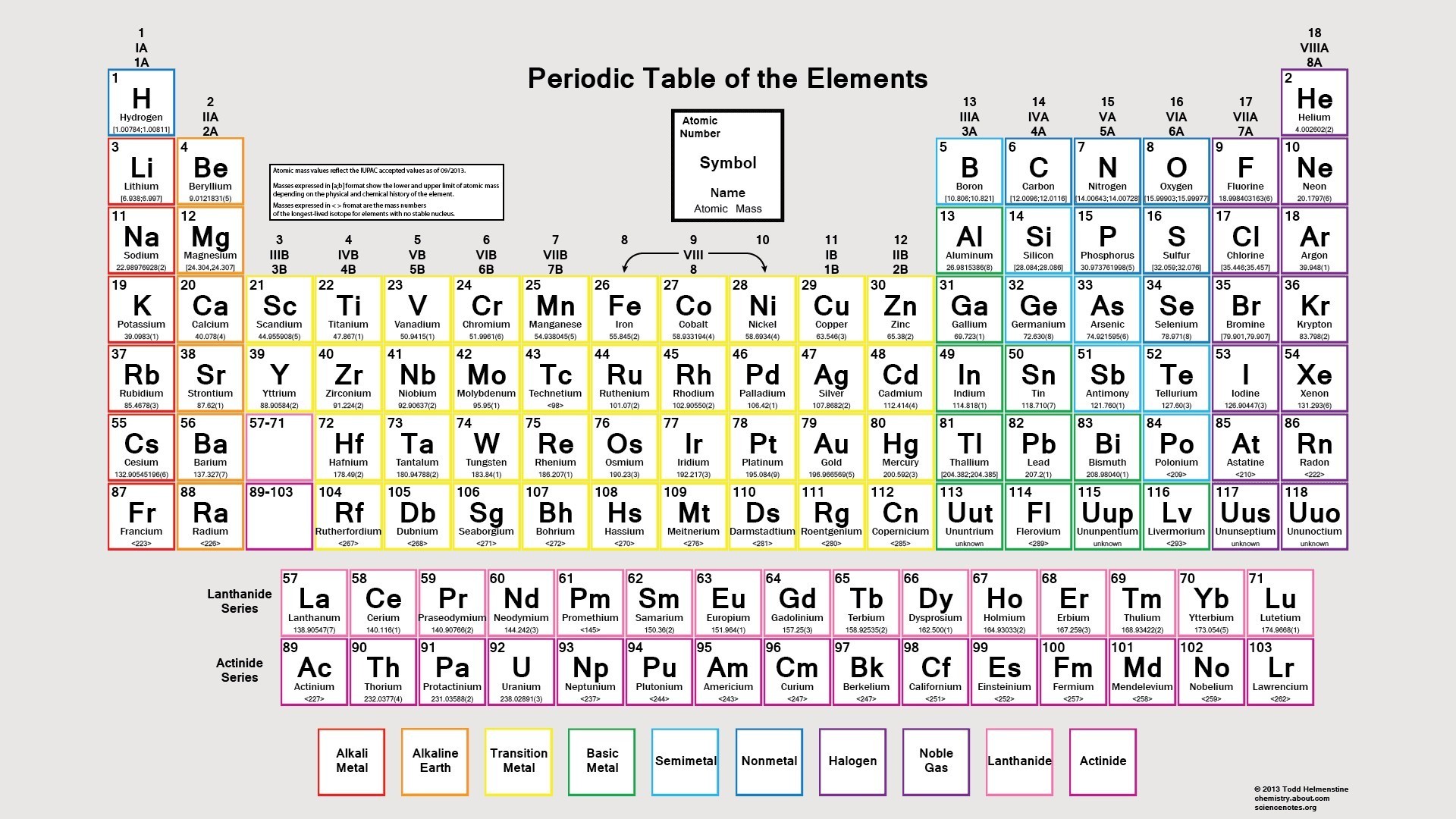
Scientists use the periodic table to quickly refer to information about an element, like atomic mass. \): Moles to Mass Conversion with Compounds The periodic table of chemical elements, often called the periodic table, organizes all discovered chemical elements in rows (called periods) and columns (called groups) according to increasing atomic number.
